Unit 33 - Acids and Bases II
Learning Outcomes:
ii. 2HCl + CaCO3 → CaCl2 + CO2+ H2O (word equation O.L.) (OC37)
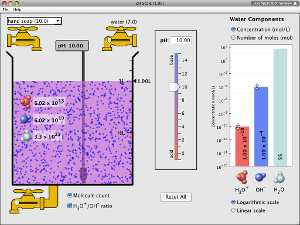
- Be able to state the names and formulae of common strong acids and bases: H2SO4, HCl, NaOH, Ca(OH)2, and understand that alkalis are soluble bases (OC35)
- Show the neutralisation of an acid with a base using an indicator (OC36)
- Understand that, when an acid reacts with a base, a salt and water are formed:
ii. 2HCl + CaCO3 → CaCl2 + CO2+ H2O (word equation O.L.) (OC37)
- Titrate HCl against NaOH, and prepare a sample of NaCl (OC38)